Mouse-derived Isograft (MDI) Models
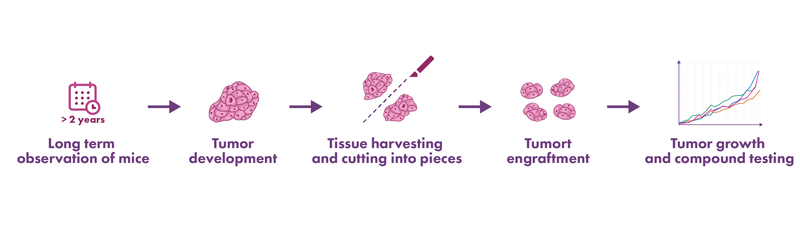
Our proprietary mouse-derived isograft tumor models (MDI) allow drug efficacy testing in a setting that recapitulates the original tumor very closely. Tumor tissue was isolated from mice with cancer, and the tissue was engrafted for propagation into other mice – also called tumor homograft model. Mouse isograft models enable the investigation of interactions between cells, stroma, and the tumor environment during cancer progression.
While cell line-derived syngeneic tumor models are based on cultured cell lines, our proprietary mouse-derived isograft (MDI) models are propagated in mice only. Hence, the major advantage of these novel spontaneous-derived or carcinogen-induced MDI tumor models is preserving the primary tumor phenotype and intratumoral immune cell populations.
- Derived from spontaneous tumors or carcinogen-induced tumors
- Allow for the identification of biomarkers
- Efficacy testing with high predictive value for patient-derived xenograft studies
- All isograft tumor model projects follow meticulous documentation per GV-Solas requirements and in line with ISO 9001 certification
- Every in vivo project is concluded with custom-tailored reports written by PhD-level medical writers
- Clients will be working with one team of scientists throughout the MDI project.
Reaction Biology has a global team of business development managers available for in depth conversations about your research needs and help determine the right models for your drug discovery project. Contact us today to inquire about our mice-derived syngeneic models or to request a quote.