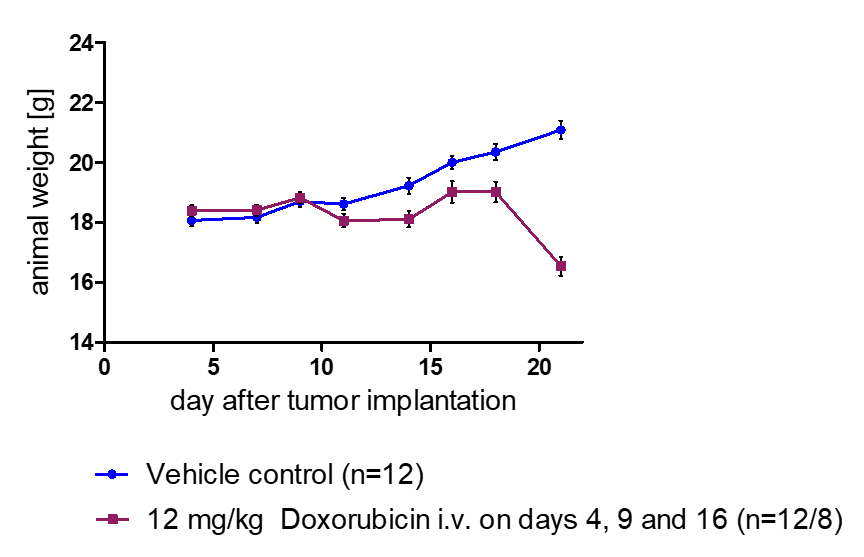
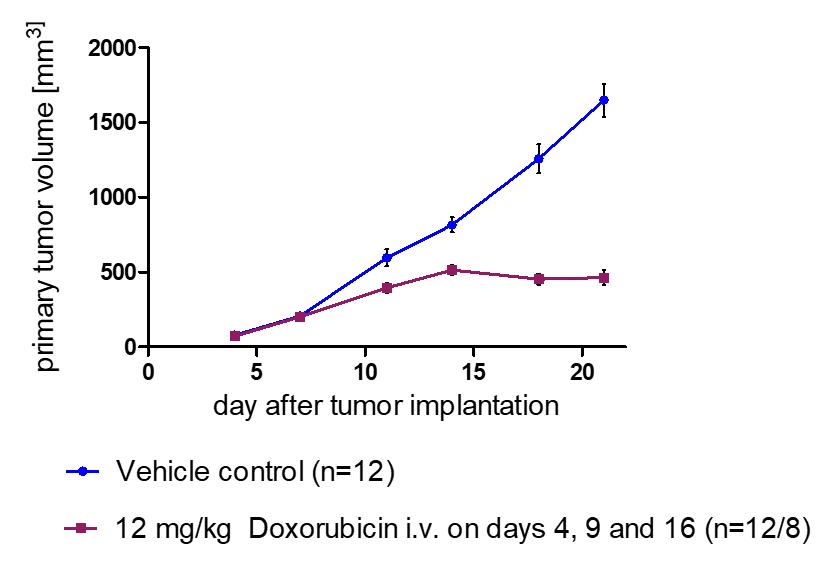
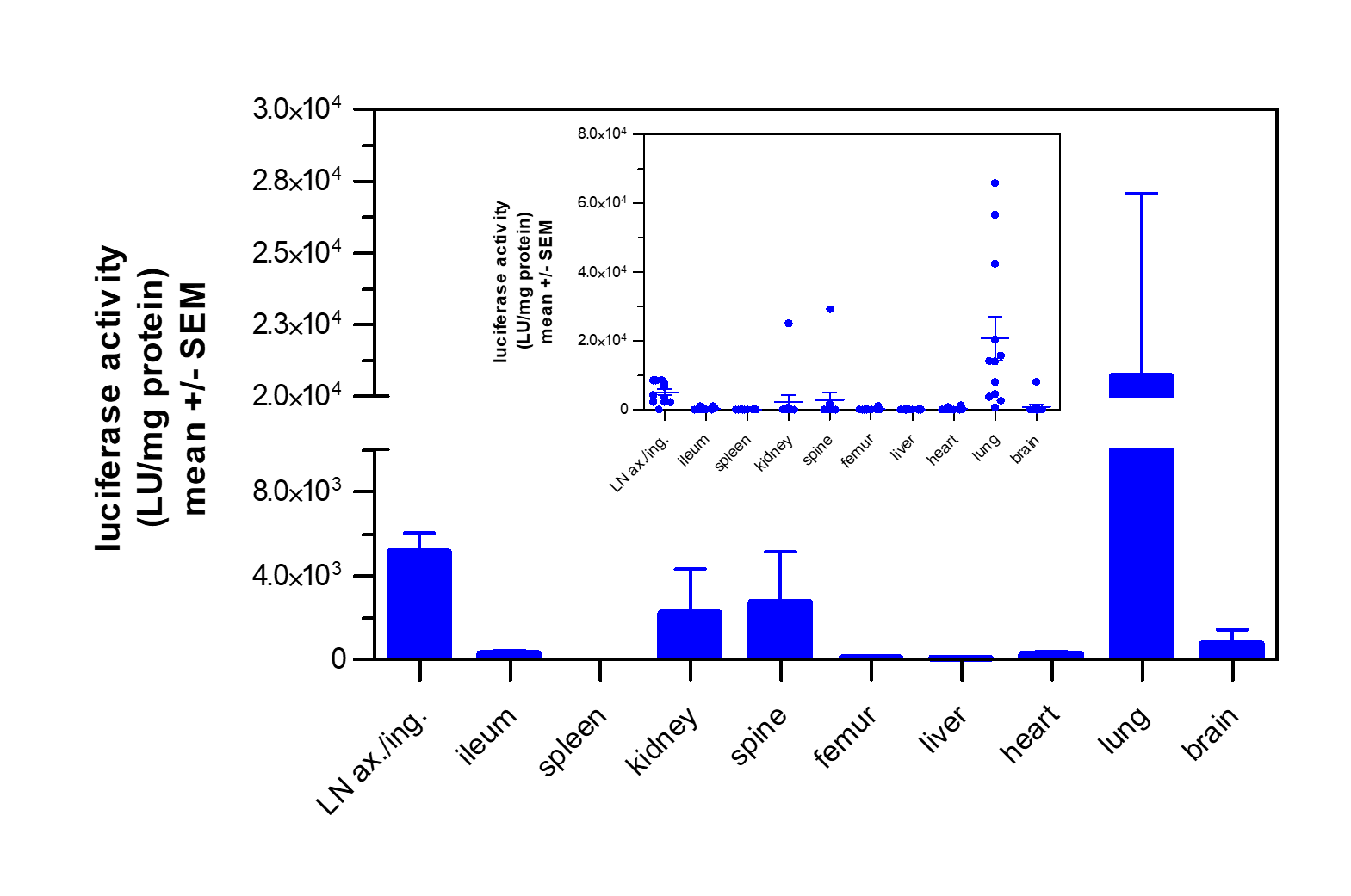
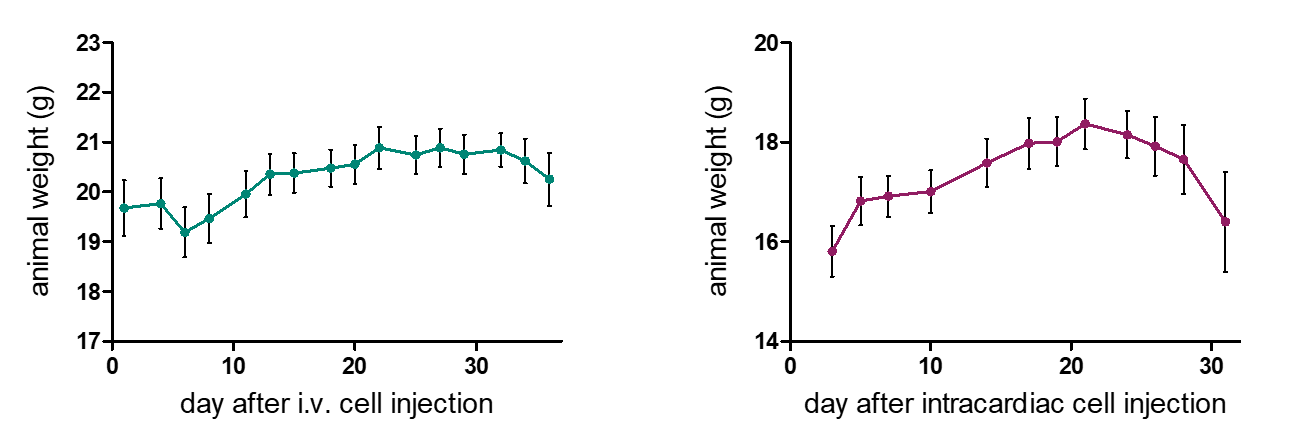In Vivo Metastasis Assays
In vivo metastasis models are designed to analyze the anti-metastatic effect of novel anti-cancer drug candidates.
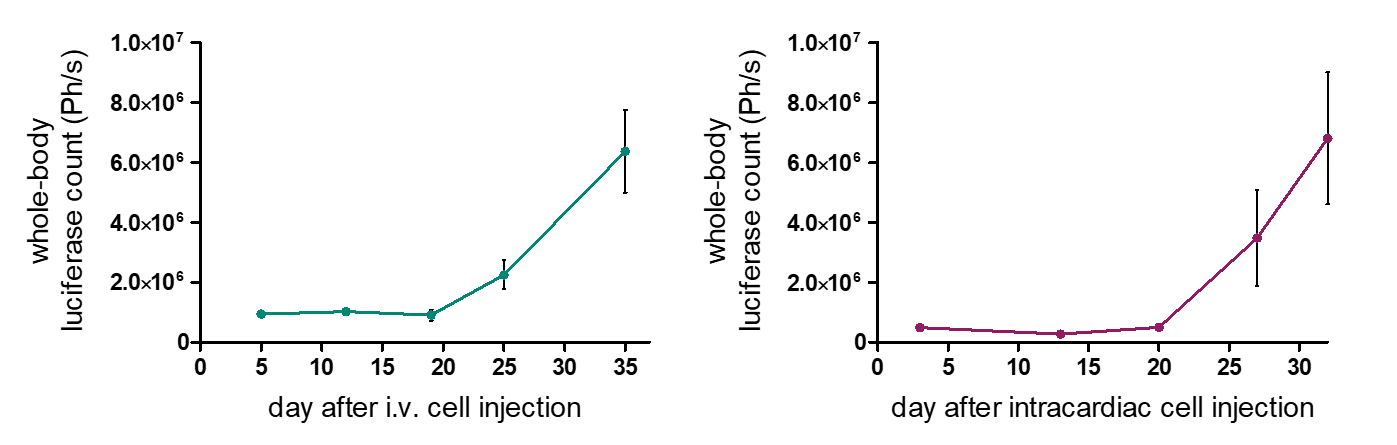
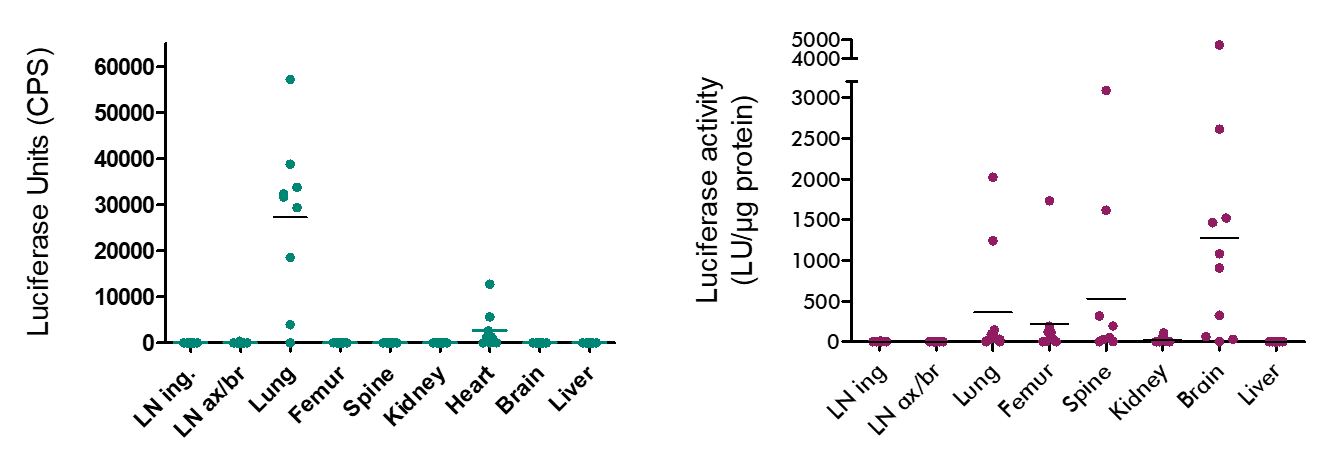
Metastasis mouse models comprise two types: 1. Orthotopic: Metastatic cells originate from the orthotopically growing tumors, followed by the migration of tumor cells to the metastasis site. 2. Disseminated: Heart or tail vein injection of tumor cells result in the establishment of metastases in the lung, bone marrow, or brain depending on the metastasis model’s tumor cell line and route of administration. The various metastasis models enable differential examination of the metastasis process.
Reaction Biology offers syngeneic and xenograft metastasis models with advanced imaging options for whole-body monitoring of metastasis.
- Whole-body imaging via IVIS allows monitoring of tumor growth kinetics and therapeutic efficacy of a drug candidate
- Models are established with different injection routes to mimic different metastasis patterns
- In vivo metastasis model study reports are written by PhD-level medical writers and are custom-tailored for each project
- Meticulous documentation in line with ISO 9001 certification and GV-Solas requirements
Reaction Biology has a team of global business development managers free to discuss in most time zones. Contact us today for inquiries about our xenograft and syngeneic metastasis models or to directly request a quote.